Covalent Bonds
Now we know that the theory of semiconductors is built upon introducing some impurity into crystalline structure of such a material as silicon (Si) or germanium (Ge).
Let's consider this crystalline structure in more details to understand its underlying principles.
According to the orbital model of an atom, electrons are rotating around a nucleus on certain orbits, each orbit representing certain energy level possessed by all electrons on it. There are certain principles related to quantum mechanics and wave theory of elementary particles that prescribe electrons to rotate only on certain distinct radiuses around a nucleus. The reason for this we will discuss when addressing the atomic structure in more details in a corresponding part of this course.
On every distinct radius the rotating electrons experience the attraction of the protons in a nucleus and repelling of other electrons rotating on the same orbit (on the same radius from a nucleus). Obviously, for every distinct radius of an orbit there is a maximum number of electrons that can rotate around a nucleus without pushing each other so strongly that one of them must jump out of the orbit to, most likely, the orbit of a larger radius or even outside of the atom.
Calculations based on principles of quantum mechanics show that this maximum number for the first (closest to a nucleus) orbit is 2, for the next - 8, next - 18 etc. The formula N=2n² where n is the orbit number and N is the maximum number of electrons on this orbit describes this in many (but not all) cases.
The atom of every element has certain number of protons in a nucleus and the same number of electrons rotating on different orbits. These orbits are packed with electrons not to exceed their maximum capacity.
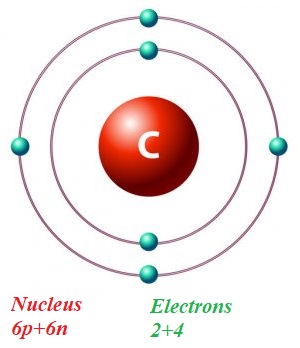
In a simplified model of an atom of carbon C12 with 6 protons and 6 neutrons in a nucleus there are 6 electrons on two orbits: 2 on the closest to a nucleus and 4 on the next one.
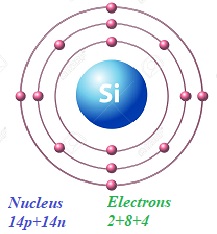
In a simplified model of an atom of silicon Si28 with 14 protons and 14 neutrons in a nucleus there are 14 electrons on three orbits: 2 on the closest to a nucleus, 8 on the next one and 4 on the outer most orbit.
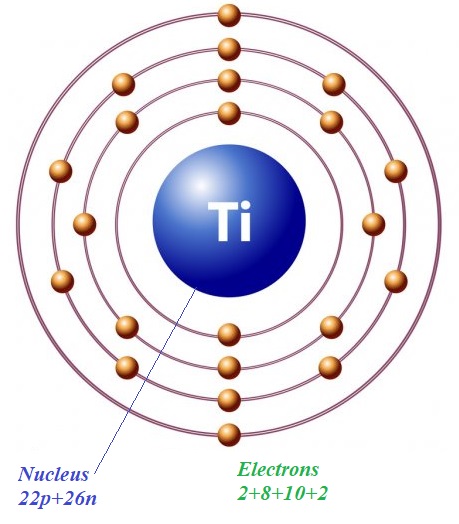
In a simplified model of an atom of titanium Ti48 with 22 protons and 26 neutrons in a nucleus there are 22 electrons on four orbits with, correspondingly, 2, 8, 10 and 2 electrons on them.
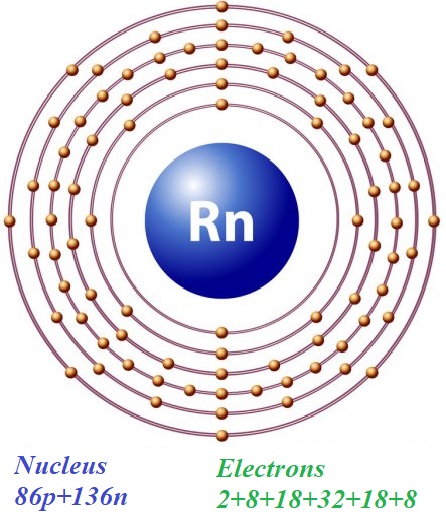
In a simplified model of an atom of radon Rn222 with 86 protons and 136 neutrons in a nucleus there are 86 electrons on five orbits with, correspondingly, 2, 8, 18, 32, 18 and 8 electrons on them.
Electrons populating the outer most orbit are called valence electrons. While in theory belonging to particular atoms, they are very active in their relationships with other atoms and their valence electrons.
In fact, valence electrons from different atoms might bond together, thus two atoms have a shared pair of electrons. The reason to this is a tendency to fill up the orbit of valence electrons. The magic number of valence electrons is 2 for atoms with only one orbit (there are only two elements with a single orbit of electrons - hydrogen and helium) or 8 for atoms with two or more orbits, so two or more atoms might come into bonding by sharing the valence electrons, thus filling all outer orbits up to a magic number of electrons (shared and not shared).
Here is an example of a molecule of methane that consists of one atom of carbon C12 (two orbits with 2 electrons on an inner orbit, not shown on a picture, and 4 valence electrons) and four atoms of hydrogen H1 (single orbit with 1 electron on it).
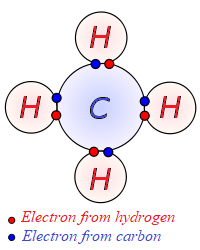
As we see, the orbit with valence electrons of an atom of carbon has now 8 electrons, 4 its own and 4 shared with atoms of hydrogen, thus filling a magic number of valence electrons. At the same time each atom of hydrogen has 2 electrons (also a magic number for an atom of hydrogen), 1 its own and one shared with an atom of carbon.
These covalent bonds between atoms are the basis for certain different atoms to combine into molecules or the same atoms to form a crystalline structure.
Consider now a crystalline structure of a silicon, our main subject of discussion.
Each atom of silicon contains 4 valence electrons on its outer (third) orbit. In three-dimensional world these electrons are positioned at the vertices of a tetrahedron with a nucleus of an atom at the center of this tetrahedron.
Four valence electrons of one atom of silicon pair with valence electrons of neighboring atoms to form a complicated three-dimensional crystalline structure that in two-dimensional representation looks as follows.
Four neighboring silicon atoms contribute to a center one their valence electrons to share (one from each neighbor), thus the center atom has a magic number of electrons on its outer orbit. Each valence electron of each atom is shared with a neighboring atom and, therefore, every atom has 8 electrons (including both its own 4 electrons and 4 from its four neighbors, one from each). This makes a strong crystalline structure of material.


No comments:
Post a Comment